To get back my youth I would do anything in the world, except take exercise, get up early, or be respectable.
Oscar Wilde, The Picture of Dorian Gray
 |
| Detail from Old woman and boy with candles (c. 1616–1617) by Peter Paul Rubens. |
EVERLASTING YOUTH is one of humanity’s perpetual aspirations. None of us are impervious to the effects of old age, either in ourselves or in those we love. Yet, more than an inescapable element of the human condition, ageing is in fact a universal biological feature of complex animals, and possibly of all life. Biologically speaking, ageing is a gradual decline in the capacity of the cells and tissues in a body to preserve their integrity and carry out their central physiological functions. The ultimate consequence of this process is the body’s inability to sustain its own existence, leading to an inevitable death from ‘old age’. Regardless of how much effort is devoted to prolonging life, humans and other animals seem to carry an intrinsic ‘expiry date’. But why should this be so? How did such an implacable force of decay come to exist, and why do we humans seem unable to vanquish it?
The question of what causes ageing, which can be traced as far back as Aristotle, is in fact composed of two very distinct questions. The first is the question of why we age: what is the ultimate biological reason for the fact that animals have never evolved the capacity to live forever? The second question is that of how we age: what are the immediate physiological processes which cause bodies gradually to decay over time? The degree to which we understand ageing may be expected to vary between these two levels of analysis — but it may come as a surprise that it should be our understanding of how we age, rather than why we age, which remains very much undeveloped. The following presents our current scientific perspective on these two dimensions of the ageing process.
Why we age: Evolutionary causes of ageing
The universality of ageing among animals was a troublesome fact to early evolutionary biologists. In the mid-nineteenth century, Charles Darwin had proposed that the biological traits of organisms were the outcome of evolution by natural selection, and therefore had probably been useful for the survival and reproduction of previous generations. How is it, then, that evolution has not crafted organisms with the clearly beneficial capacity to maintain their youth indefinitely?
The first evolutionary explanation of ageing was proposed by the nineteenth-century biologist August Weismann. An early supporter of Darwin’s ideas, Weismann was a key figure in the development of early theories of biological heredity. To him, the evolutionary paradox of ageing could be resolved if one assumed that an animal’s longevity is indeed the product of natural selection — but not because of any benefit to the animal itself, but rather to the species as a whole. He proposed that the duration of life — the lifespan — has evolved to an optimal value which spares the population from being smothered by a preponderance of old individuals. In Weismann’s account, ageing is therefore a death mechanism explicitly evolved for the purging of older, less competitive generations, enabling the success of younger individuals. Remarkably, this theory was in fact a Darwinian makeover of the views of the ancient Roman poet and philosopher Lucretius.
Weismann’s explanation of ageing, although intuitively cogent, was found by later evolutionary biologists to be flawed. For one thing, the argument that older individuals should be purged because they are less fit than younger ones immediately invokes an assumption that individuals experience physiological ageing. But to infer the evolutionary origins of ageing, we must begin with a population whose individuals do not age, and thus can only die through extrinsic forces such as predation, infection, starvation or accident. In such a population, there is no reason to assume that older individuals should be at a disadvantage — if anything, the fact that they have survived for longer implies that they are, on average, better survivors. Moreover, older individuals should have amassed precious expertise in the manoeuvres and tactics of living, such that they should offer formidable competition to youngsters. Therefore, without the assumption of an ageing process, the death of older individuals cannot easily be defended as of benefit to the species.
Another powerful argument against Weismann’s theory is the now-established fact that traits which benefit the collective at the expense of the individual are evolutionarily unstable. In most situations, natural selection operates overwhelmingly at the level of the individual: if one deer is, for instance, able to outrun the others, it will be less likely to be preyed upon, and hence more likely to leave offspring, which will inherit its superior speed. In the same manner, if a species were to evolve an ageing process that were beneficial to the species but disadvantageous to the individual, then any individual happening to age more slowly than the rest would be at a considerable advantage, just like the faster-running deer, and so this trait would be favoured by natural selection. Ageing therefore cannot have evolved for the sole benefit of the species; if Weismann appears here to have misjudged the implications of Darwin’s theory, it may be said in his defence that Darwin himself would have fared no better. It is only after one and a half centuries of thought that we have come to understand ageing not as a consequence of the direct action of natural selection — but rather of its failure.
One of the earliest hints at the concept which underlies modern evolutionary theories of ageing was advanced by the influential mathematical geneticist JBS Haldane. During an inspired series of lectures in 1940, Haldane noted in passing that natural selection should have little power to suppress a deleterious trait if such a trait only manifests itself late in life. To see why this is the case, let us consider Haldane’s case of interest — Huntington’s disease. Despite its devastating and fatal effects, this degenerative condition typically has its onset after the age of thirty, and hence has little impact on a person’s ability to have children. By the time the disease is diagnosed, the patient’s children may already have inherited the responsible gene. Haldane correctly saw this as the reason why such a pernicious gene has not been purged by evolution. The impact of Huntington’s disease is confined to adulthood, a period of life in which the strength of natural selection declines dramatically, since reproduction has already taken place. This period is now termed the ‘selection shadow’, because biological effects within it are effectively ‘out of sight’ for evolution.
The concept of the selection shadow was first developed into a complete theory of ageing by the Nobel laureate Sir Peter Medawar, who in the 1950s attempted to explain ageing as the combined effect of a collection of ‘mutant genes’ — altered versions of ‘normal’ genes — whose effects only arise late in life. Just as in the case of Huntington’s disease, age-related conditions such as cataracts, arthritis and osteoporosis have a late onset and no impact on reproduction, which precludes natural selection from weeding off the implicated ‘mutant genes’. A large number of these problematic genes will therefore accumulate in the ‘shadow’ of selection, their effects amalgamating into what we call ageing. Medawar also grasped the significance of extrinsic mortality, that is, the rate of death from environmental forces such as predation: the later in life the effects of a gene are realised, the less individuals will remain alive to experience them. Thus, a gene which contributes to prolonging the health of heart muscle for many decades may be very beneficial to an elephant, but it is of no use to a mouse that will almost certainly be preyed upon before the age of two.
Building on Medawar’s work, a later theory proposed that ageing may arise from genes which not only have negative effects in old age, but also have beneficial effects in youth, when natural selection is at its strongest. In this theory, ageing would be a detrimental late by-product of processes which have evolved because they are beneficial earlier in life. The current scientific consensus is that each of these two theories is probably correct in some cases, such that certain components of ageing have arisen through accumulation of purely detrimental mutant genes, while others are late side-effects of advantageous genes.
An important aspect of these two evolutionary theories is that they define ageing as the result of the inability of natural selection to maintain physiological integrity for longer than is actually useful ‘in the wild’. The key insight is that it is not evolutionarily advantageous to live longer than we do, because our species has evolved so that we are able to develop and reproduce long before our bodies succumb to age. Furthermore, because the wild environment of early humans made it very unlikely for them to survive as long as we do, there has been no evolutionary need for greater longevity. Notably, our evolutionary explanation of ageing, which is theoretically and empirically well supported, does not depend on which specific physiological mechanisms are responsible for ageing. In other words, we certainly understand why the process of ageing has evolved in the first place; the scene is rather different, however, when it comes to the question of how this process unfolds in organisms.
How we age: Mechanistic causes of ageing
Luckily for junior scientists, our mechanistic theories of ageing are much more abundant and less clearly supported than our evolutionary theories. Perhaps the most immediate question regarding the actual process of ageing is whether it results from a single physiological mechanism, or from multiple mechanisms whose effects are roughly synchronised. Given the conclusion that ageing is a consequence of the ineffectiveness of natural selection, it follows that it must come about through multiple, possibly many, unrelated mechanisms.
As a crude analogy, let us imagine owning a car in a very unsafe city, where vehicles are constantly being stolen or damaged. In such circumstances, we should be wise to buy a cheap car which might last a few years, and to spend as little as possible in maintenance, as otherwise the return on our investment may never materialise. Nevertheless, if by a stroke of fortune, we find ourselves owning the same car after a good number of years, we should expect it to come apart by virtue of its being cheap and poorly maintained. This analogy unflatteringly exposes the ultimate reason for ageing — insufficient quality and care — yet it sheds no light as to which of the car’s components is expected to fail first. Given that the car’s decay is caused by deficient maintenance, we might expect multiple of its components to misbehave with increasing frequency, up to the point where the machine as a whole cannot function. Moreover, different processes might be responsible for each component’s failure: the transmission may expire out of sheer friction, while the pistons might succumb to soot. Hence, even though the ultimate cause of ageing may be universal, the processes immediately involved are manifold.
As suggested by this analogy, current research on ageing focuses on the challenging task of establishing which physiological processes contribute to ageing, and how significant each is. A large number of distinct processes have indeed been proposed as mechanistic causes of ageing. Among the most interesting of these are ‘nutrient signalling pathways’, which are functional networks of molecules responsible for transmitting the physiological signals produced when we acquire nutrients. The most popular molecule in this network is insulin, essential for the regulation of blood glucose levels. Yet in addition to the well-known relationship between deficient insulin signalling and diabetes, it has been found that interventions which interfere with nutrient signalling can considerably prolong the lifespan of many species, both vertebrate and invertebrate. For instance, a treatment known as ‘dietary restriction’, whereby the supply of food (or of certain nutrients) is permanently reduced, is considered the most reliable way of extending animal lifespan. Furthermore, the deactivation of certain nutrient signalling genes, by either mutation or pharmacological treatment, produces similar effects to those of dietary restriction. In the 1990s, Cynthia Kenyon and her colleagues discovered that mutations in such a gene led to a doubling of lifespan in nematode worms, a finding followed by similar reports in fruit flies by the groups of Dame Linda Partridge and Marc Tatar. On the other hand, nutrient signalling also regulates body growth and development, and animals subjected to these life-prolonging interventions tend to be stunted and ill-developed. Interestingly, although the network of effects whereby nutrient signalling modulates development and longevity is not yet fully characterised, it is believed to be the reason why smaller dog breeds live longer than larger ones.
A second leading candidate among possible mechanisms of ageing is molecular damage. Cells are constantly exposed to many kinds of chemical damage, which can alter their constituent molecules and impair the efficiency of cellular processes. The types of molecules subject to such damage include proteins (which are both the cell’s building blocks and its working tools) and DNA (which carries the organism’s genetic information, including the instructions for protein synthesis). One extensively studied type of DNA modification with potential roles in ageing is the shortening of telomeres — long stretches of DNA which are placed at the ends of chromosomes to protect them from fraying, like the aglet in a shoelace. Telomeres are slightly shortened every time a cell divides into two new cells, and eventually become too short to allow further cell division, which is thought to be an important barrier against the emergence of cancer — but might also be a cause of ageing. Recently, the biologist María Blasco and her team reported the striking finding that the rate of telomere shortening in a species is related to its lifespan, such that telomeres erode faster in shorter-lived species. Nevertheless, this relationship is obscured by the fact that shorter-lived species also tend to be smaller, and body size itself is thought to influence many aspects of animal physiology.
The question of what causes ageing, which can be traced as far back as Aristotle, is in fact composed of two very distinct questions. The first is the question of why we age: what is the ultimate biological reason for the fact that animals have never evolved the capacity to live forever? The second question is that of how we age: what are the immediate physiological processes which cause bodies gradually to decay over time? The degree to which we understand ageing may be expected to vary between these two levels of analysis — but it may come as a surprise that it should be our understanding of how we age, rather than why we age, which remains very much undeveloped. The following presents our current scientific perspective on these two dimensions of the ageing process.
Why we age: Evolutionary causes of ageing
The universality of ageing among animals was a troublesome fact to early evolutionary biologists. In the mid-nineteenth century, Charles Darwin had proposed that the biological traits of organisms were the outcome of evolution by natural selection, and therefore had probably been useful for the survival and reproduction of previous generations. How is it, then, that evolution has not crafted organisms with the clearly beneficial capacity to maintain their youth indefinitely?
The first evolutionary explanation of ageing was proposed by the nineteenth-century biologist August Weismann. An early supporter of Darwin’s ideas, Weismann was a key figure in the development of early theories of biological heredity. To him, the evolutionary paradox of ageing could be resolved if one assumed that an animal’s longevity is indeed the product of natural selection — but not because of any benefit to the animal itself, but rather to the species as a whole. He proposed that the duration of life — the lifespan — has evolved to an optimal value which spares the population from being smothered by a preponderance of old individuals. In Weismann’s account, ageing is therefore a death mechanism explicitly evolved for the purging of older, less competitive generations, enabling the success of younger individuals. Remarkably, this theory was in fact a Darwinian makeover of the views of the ancient Roman poet and philosopher Lucretius.
Weismann’s explanation of ageing, although intuitively cogent, was found by later evolutionary biologists to be flawed. For one thing, the argument that older individuals should be purged because they are less fit than younger ones immediately invokes an assumption that individuals experience physiological ageing. But to infer the evolutionary origins of ageing, we must begin with a population whose individuals do not age, and thus can only die through extrinsic forces such as predation, infection, starvation or accident. In such a population, there is no reason to assume that older individuals should be at a disadvantage — if anything, the fact that they have survived for longer implies that they are, on average, better survivors. Moreover, older individuals should have amassed precious expertise in the manoeuvres and tactics of living, such that they should offer formidable competition to youngsters. Therefore, without the assumption of an ageing process, the death of older individuals cannot easily be defended as of benefit to the species.
Another powerful argument against Weismann’s theory is the now-established fact that traits which benefit the collective at the expense of the individual are evolutionarily unstable. In most situations, natural selection operates overwhelmingly at the level of the individual: if one deer is, for instance, able to outrun the others, it will be less likely to be preyed upon, and hence more likely to leave offspring, which will inherit its superior speed. In the same manner, if a species were to evolve an ageing process that were beneficial to the species but disadvantageous to the individual, then any individual happening to age more slowly than the rest would be at a considerable advantage, just like the faster-running deer, and so this trait would be favoured by natural selection. Ageing therefore cannot have evolved for the sole benefit of the species; if Weismann appears here to have misjudged the implications of Darwin’s theory, it may be said in his defence that Darwin himself would have fared no better. It is only after one and a half centuries of thought that we have come to understand ageing not as a consequence of the direct action of natural selection — but rather of its failure.
One of the earliest hints at the concept which underlies modern evolutionary theories of ageing was advanced by the influential mathematical geneticist JBS Haldane. During an inspired series of lectures in 1940, Haldane noted in passing that natural selection should have little power to suppress a deleterious trait if such a trait only manifests itself late in life. To see why this is the case, let us consider Haldane’s case of interest — Huntington’s disease. Despite its devastating and fatal effects, this degenerative condition typically has its onset after the age of thirty, and hence has little impact on a person’s ability to have children. By the time the disease is diagnosed, the patient’s children may already have inherited the responsible gene. Haldane correctly saw this as the reason why such a pernicious gene has not been purged by evolution. The impact of Huntington’s disease is confined to adulthood, a period of life in which the strength of natural selection declines dramatically, since reproduction has already taken place. This period is now termed the ‘selection shadow’, because biological effects within it are effectively ‘out of sight’ for evolution.
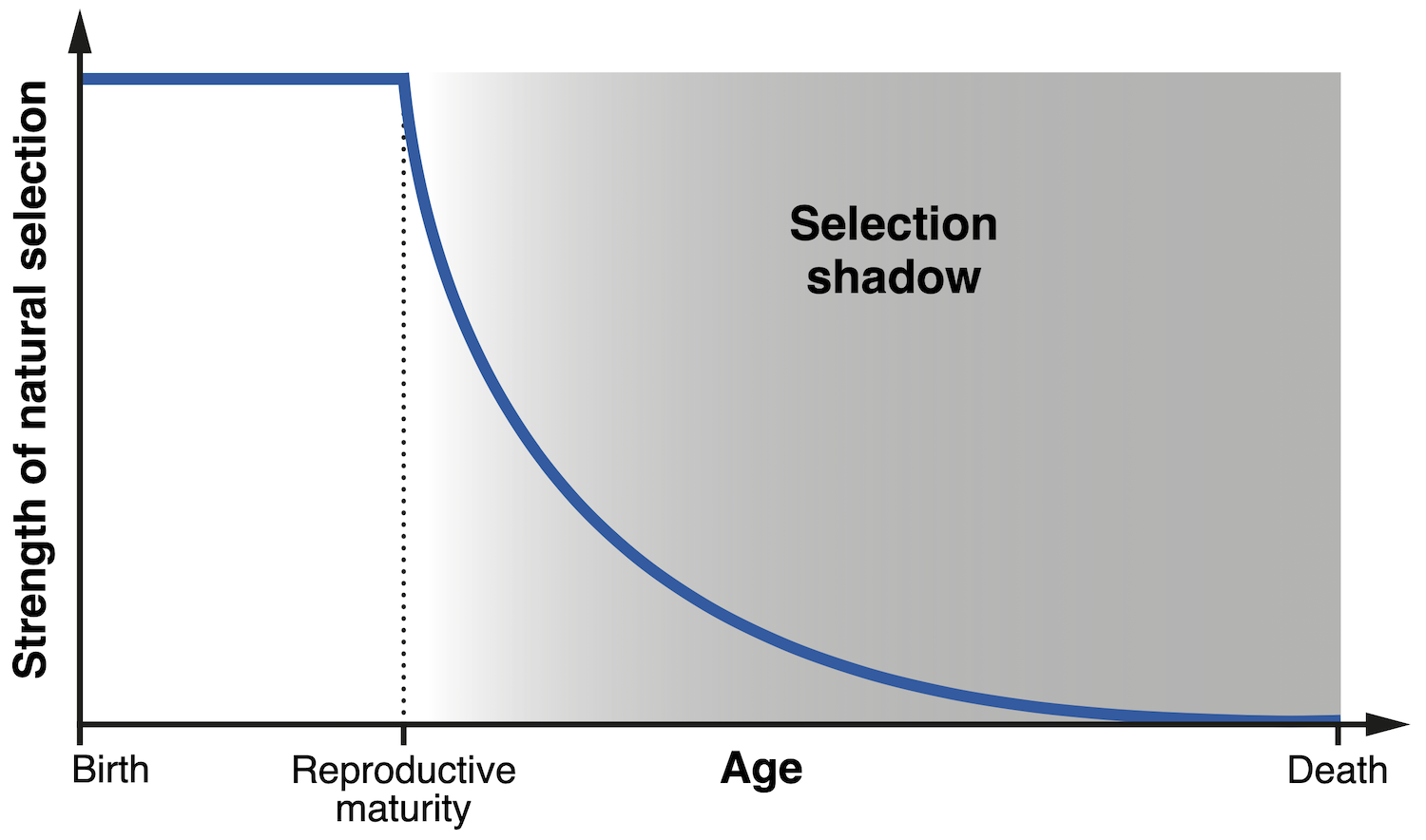 |
| Diagram illustrating the concept of the ‘selection shadow’, referring to the progressive decline in the strength of natural selection after the age of reproductive maturity (Credit: A Baez-Ortega). |
The concept of the selection shadow was first developed into a complete theory of ageing by the Nobel laureate Sir Peter Medawar, who in the 1950s attempted to explain ageing as the combined effect of a collection of ‘mutant genes’ — altered versions of ‘normal’ genes — whose effects only arise late in life. Just as in the case of Huntington’s disease, age-related conditions such as cataracts, arthritis and osteoporosis have a late onset and no impact on reproduction, which precludes natural selection from weeding off the implicated ‘mutant genes’. A large number of these problematic genes will therefore accumulate in the ‘shadow’ of selection, their effects amalgamating into what we call ageing. Medawar also grasped the significance of extrinsic mortality, that is, the rate of death from environmental forces such as predation: the later in life the effects of a gene are realised, the less individuals will remain alive to experience them. Thus, a gene which contributes to prolonging the health of heart muscle for many decades may be very beneficial to an elephant, but it is of no use to a mouse that will almost certainly be preyed upon before the age of two.
Building on Medawar’s work, a later theory proposed that ageing may arise from genes which not only have negative effects in old age, but also have beneficial effects in youth, when natural selection is at its strongest. In this theory, ageing would be a detrimental late by-product of processes which have evolved because they are beneficial earlier in life. The current scientific consensus is that each of these two theories is probably correct in some cases, such that certain components of ageing have arisen through accumulation of purely detrimental mutant genes, while others are late side-effects of advantageous genes.
An important aspect of these two evolutionary theories is that they define ageing as the result of the inability of natural selection to maintain physiological integrity for longer than is actually useful ‘in the wild’. The key insight is that it is not evolutionarily advantageous to live longer than we do, because our species has evolved so that we are able to develop and reproduce long before our bodies succumb to age. Furthermore, because the wild environment of early humans made it very unlikely for them to survive as long as we do, there has been no evolutionary need for greater longevity. Notably, our evolutionary explanation of ageing, which is theoretically and empirically well supported, does not depend on which specific physiological mechanisms are responsible for ageing. In other words, we certainly understand why the process of ageing has evolved in the first place; the scene is rather different, however, when it comes to the question of how this process unfolds in organisms.
How we age: Mechanistic causes of ageing
Luckily for junior scientists, our mechanistic theories of ageing are much more abundant and less clearly supported than our evolutionary theories. Perhaps the most immediate question regarding the actual process of ageing is whether it results from a single physiological mechanism, or from multiple mechanisms whose effects are roughly synchronised. Given the conclusion that ageing is a consequence of the ineffectiveness of natural selection, it follows that it must come about through multiple, possibly many, unrelated mechanisms.
As a crude analogy, let us imagine owning a car in a very unsafe city, where vehicles are constantly being stolen or damaged. In such circumstances, we should be wise to buy a cheap car which might last a few years, and to spend as little as possible in maintenance, as otherwise the return on our investment may never materialise. Nevertheless, if by a stroke of fortune, we find ourselves owning the same car after a good number of years, we should expect it to come apart by virtue of its being cheap and poorly maintained. This analogy unflatteringly exposes the ultimate reason for ageing — insufficient quality and care — yet it sheds no light as to which of the car’s components is expected to fail first. Given that the car’s decay is caused by deficient maintenance, we might expect multiple of its components to misbehave with increasing frequency, up to the point where the machine as a whole cannot function. Moreover, different processes might be responsible for each component’s failure: the transmission may expire out of sheer friction, while the pistons might succumb to soot. Hence, even though the ultimate cause of ageing may be universal, the processes immediately involved are manifold.
As suggested by this analogy, current research on ageing focuses on the challenging task of establishing which physiological processes contribute to ageing, and how significant each is. A large number of distinct processes have indeed been proposed as mechanistic causes of ageing. Among the most interesting of these are ‘nutrient signalling pathways’, which are functional networks of molecules responsible for transmitting the physiological signals produced when we acquire nutrients. The most popular molecule in this network is insulin, essential for the regulation of blood glucose levels. Yet in addition to the well-known relationship between deficient insulin signalling and diabetes, it has been found that interventions which interfere with nutrient signalling can considerably prolong the lifespan of many species, both vertebrate and invertebrate. For instance, a treatment known as ‘dietary restriction’, whereby the supply of food (or of certain nutrients) is permanently reduced, is considered the most reliable way of extending animal lifespan. Furthermore, the deactivation of certain nutrient signalling genes, by either mutation or pharmacological treatment, produces similar effects to those of dietary restriction. In the 1990s, Cynthia Kenyon and her colleagues discovered that mutations in such a gene led to a doubling of lifespan in nematode worms, a finding followed by similar reports in fruit flies by the groups of Dame Linda Partridge and Marc Tatar. On the other hand, nutrient signalling also regulates body growth and development, and animals subjected to these life-prolonging interventions tend to be stunted and ill-developed. Interestingly, although the network of effects whereby nutrient signalling modulates development and longevity is not yet fully characterised, it is believed to be the reason why smaller dog breeds live longer than larger ones.
A second leading candidate among possible mechanisms of ageing is molecular damage. Cells are constantly exposed to many kinds of chemical damage, which can alter their constituent molecules and impair the efficiency of cellular processes. The types of molecules subject to such damage include proteins (which are both the cell’s building blocks and its working tools) and DNA (which carries the organism’s genetic information, including the instructions for protein synthesis). One extensively studied type of DNA modification with potential roles in ageing is the shortening of telomeres — long stretches of DNA which are placed at the ends of chromosomes to protect them from fraying, like the aglet in a shoelace. Telomeres are slightly shortened every time a cell divides into two new cells, and eventually become too short to allow further cell division, which is thought to be an important barrier against the emergence of cancer — but might also be a cause of ageing. Recently, the biologist María Blasco and her team reported the striking finding that the rate of telomere shortening in a species is related to its lifespan, such that telomeres erode faster in shorter-lived species. Nevertheless, this relationship is obscured by the fact that shorter-lived species also tend to be smaller, and body size itself is thought to influence many aspects of animal physiology.
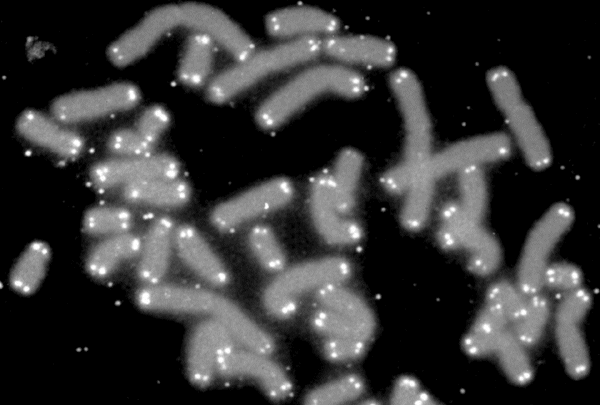 |
| Fluorescence microscopy image showing the location of telomeres (white) at the ends of human chromosomes (grey). Telomeres preserve the integrity of DNA inside chromosomes, and their shortening over time has been proposed as a cause of ageing (Credit: NASA/Wikimedia Commons, public domain). |
Working with Alex Cagan, Iñigo Martincorena and other researchers at the Wellcome Sanger Institute, I recently explored the relationship between animal lifespan and another common form of DNA modification — somatic mutations. This term refers to the changes that accrue in our DNA over time; such mutations are not present initially in any of our cells, but are acquired by individual cells as our bodies grow and age. Somatic mutations were first hypothesised to contribute to ageing in the 1960s, but their exact role remains elusive. Cagan and I characterised the rate of mutation across sixteen species of mammals, from mice to giraffes, and found a very similar relationship to that described for telomeres: shorter-lived species mutate faster than longer-lived ones, such that a mouse cell acquires as many mutations in two years as a human cell might do in eighty. Crucially, we determined this result to be unaffected by the relationship between longevity and body size: at least in mammals, the mutation rate can be used to predict a species’ lifespan, regardless of its size. The fact that the rates of different forms of molecular damage present similar relationships with lifespan suggests — but does not prove — that these forms of damage may be involved in ageing.
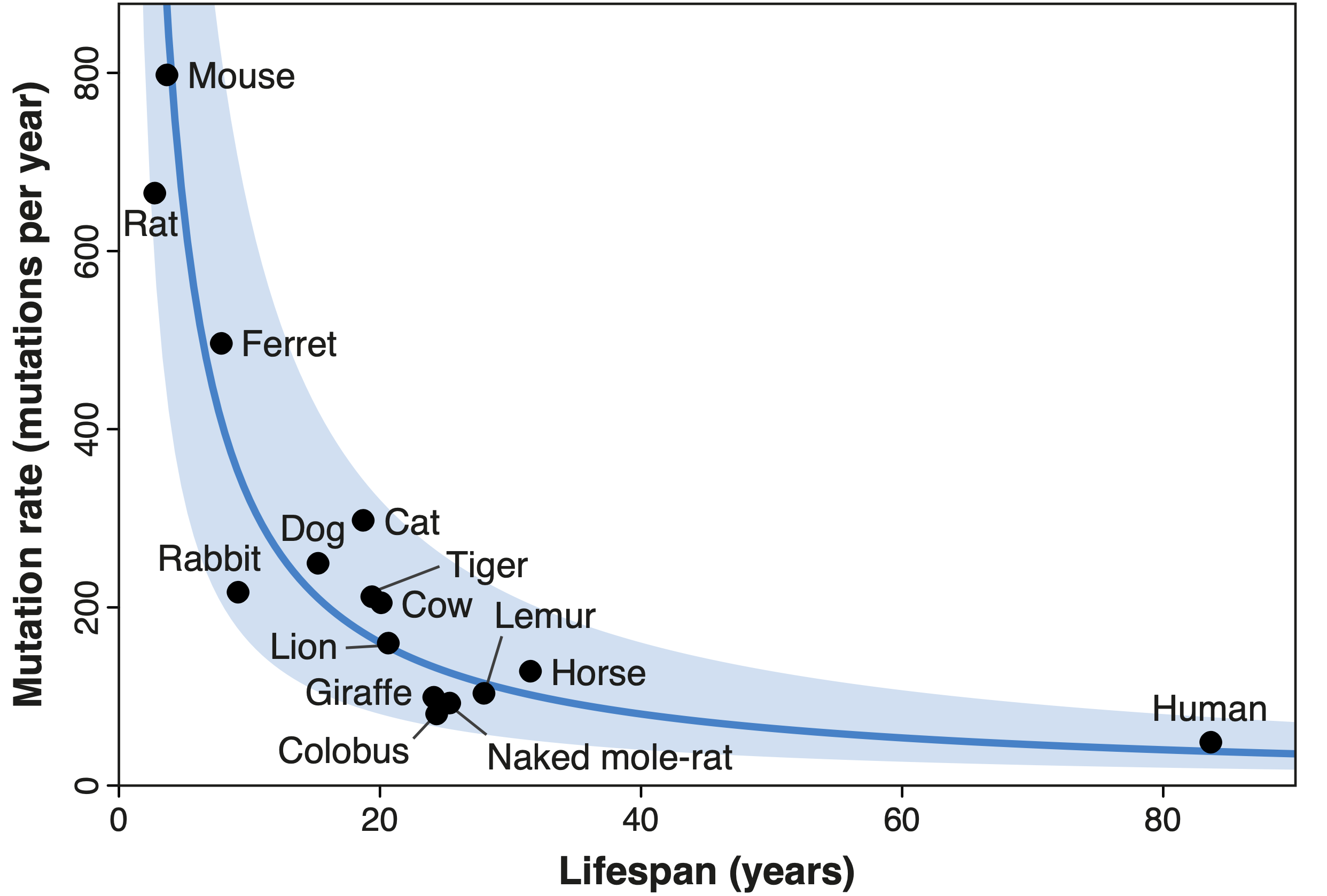 |
| Diagram showing the inverse relationship between lifespan and the rate of somatic mutation in 16 species of mammals. The mutation rate of each species is inversely proportional to its lifespan, such that all species carry a similar number of mutations in their cells’ DNA at the end of their respective lifespans. This relationship is indicated by the blue line, with the shaded area marking a two-fold deviation from this line (Source: Cagan, Baez-Ortega et al., 2022). |
It might seem inconsistent that processes as unrelated as nutrient signalling and molecular damage might all contribute to ageing. But these processes are not so distant when viewed in the light of a theory known as the ‘disposable soma’ theory of ageing. According to this, the physiology of complex organisms includes a central energy trade-off, such that the energy acquired from food is distributed between the processes of somatic maintenance (the preservation of the body via repair of molecular damage) and reproduction (the preservation of genes via their transmission to offspring). Rather than grappling with the evolutionary origin of ageing, this theory provides a framework for its physiological regulation. Because our body (the ‘soma’) is ultimately perishable, the energy trade-off between maintenance and reproduction has presumably been optimised by evolution to favour the expensive process of reproduction in times of nutrient abundance, and to promote maintenance instead when nutrients are limited. It is thus possible that nutrient signalling disruption modifies the speed of ageing by interfering with the ‘gauge’ of this energy allocation system, whereas molecular damage may simply be the force which opposes somatic maintenance processes. Despite the remarkable coherence of the disposable soma theory, the evidence for the existence of a universal energy trade-off in animals is currently inconclusive. It is possible that, like so much else in biology, energy trade-offs are crucial but not universal: they might be relevant only for some species, or in certain organs, or at particular periods in life. Even in this time of unparalleled scientific progress, an immensity of knowledge remains to be discovered regarding the physiological processes involved in ageing.
The battle against ageing
Since the days of Darwin and Weismann, we have come to understand ageing not as a death force evolved for the benefit of the species, but rather as an inextricable consequence of the manner in which evolution works. Animal bodies have not evolved to live forever, but to succeed in surviving and reproducing amidst a ruthless environment. The biology of our bodies is such as it is precisely because our ancestors managed to succeed in these tasks, not because they managed to live forever.
Whatever the causes of ageing, the essential question for humanity is whether we shall ever be able to throttle them — perhaps not with a view to living forever, but at least to enjoying longer-lasting health and a happier old age. It seems clear that this target will remain out of reach so long as we fail to understand what exactly ‘ageing’ means at the molecular level. Someday we might gain the power to manipulate the processes by which our bodies fend off the effects of time, or even to combat such effects directly; we may finally be able to subdue and domesticate the process of ageing. But such miracles lie still beyond the horizon, and for years to come we must keep drawing on the power of conventional medicine to manage individual age-related conditions.
When it comes to growing older, the personal theory of the essayist, poet and former Master of Magdalene College, AC Benson, may be more helpful than those discussed here: ‘I have a theory that one ought to grow older in a tranquil and appropriate way, that one ought to be perfectly contented with one’s time of life, that amusements and pursuits ought to alter naturally and easily, and not be regretfully abandoned’. Too modest a theory, perhaps; he goes on to concede that ‘It is easier said than done’. Yet, even as we feel the gentle, impassive slipping away of youth between our fingers, we should be wise to keep in mind the words of Longfellow:
The battle against ageing
Since the days of Darwin and Weismann, we have come to understand ageing not as a death force evolved for the benefit of the species, but rather as an inextricable consequence of the manner in which evolution works. Animal bodies have not evolved to live forever, but to succeed in surviving and reproducing amidst a ruthless environment. The biology of our bodies is such as it is precisely because our ancestors managed to succeed in these tasks, not because they managed to live forever.
Whatever the causes of ageing, the essential question for humanity is whether we shall ever be able to throttle them — perhaps not with a view to living forever, but at least to enjoying longer-lasting health and a happier old age. It seems clear that this target will remain out of reach so long as we fail to understand what exactly ‘ageing’ means at the molecular level. Someday we might gain the power to manipulate the processes by which our bodies fend off the effects of time, or even to combat such effects directly; we may finally be able to subdue and domesticate the process of ageing. But such miracles lie still beyond the horizon, and for years to come we must keep drawing on the power of conventional medicine to manage individual age-related conditions.
When it comes to growing older, the personal theory of the essayist, poet and former Master of Magdalene College, AC Benson, may be more helpful than those discussed here: ‘I have a theory that one ought to grow older in a tranquil and appropriate way, that one ought to be perfectly contented with one’s time of life, that amusements and pursuits ought to alter naturally and easily, and not be regretfully abandoned’. Too modest a theory, perhaps; he goes on to concede that ‘It is easier said than done’. Yet, even as we feel the gentle, impassive slipping away of youth between our fingers, we should be wise to keep in mind the words of Longfellow:
For age is opportunity no less
Than youth itself, though in another dress,
And as the evening twilight fades away
The sky is filled with stars, invisible by day.
References
Weismann, A. ‘The duration of life’ (1881). In Essays Upon Heredity and Kindred Biological Problems (tr. Poulton, EB, Schönland, S, Shipley, AE). Clarendon, 1889.
Haldane, JBS. New Paths in Genetics. Allen & Unwin, 1941.
Kenyon, C, Chang, J et al. A C. elegans mutant that lives twice as long as wild type. Nature, 1993.
Hughes, KA, Reynolds, RM. Evolutionary and mechanistic theories of aging. Annual Review of Entomology, 2005.
Kirkwood, TBL. Understanding the odd science of aging. Cell, 2005.
Flatt, T, Partridge, L. Horizons in the evolution of aging. BMC Biology, 2018.
Whittemore, K, Vera, E et al. Telomere shortening rate predicts species life span. Proceedings of the National Academy of Sciences, 2019.
Cagan, A, Baez-Ortega, A et al. Somatic mutation rates scale with lifespan across mammals. Nature, 2022.
This article was originally published in the 2021–22 Magdalene College Magazine.
The author is grateful to James Raven and Aude Fitzsimons for their comments on the original manuscript.
Weismann, A. ‘The duration of life’ (1881). In Essays Upon Heredity and Kindred Biological Problems (tr. Poulton, EB, Schönland, S, Shipley, AE). Clarendon, 1889.
Haldane, JBS. New Paths in Genetics. Allen & Unwin, 1941.
Kenyon, C, Chang, J et al. A C. elegans mutant that lives twice as long as wild type. Nature, 1993.
Hughes, KA, Reynolds, RM. Evolutionary and mechanistic theories of aging. Annual Review of Entomology, 2005.
Kirkwood, TBL. Understanding the odd science of aging. Cell, 2005.
Flatt, T, Partridge, L. Horizons in the evolution of aging. BMC Biology, 2018.
Whittemore, K, Vera, E et al. Telomere shortening rate predicts species life span. Proceedings of the National Academy of Sciences, 2019.
Cagan, A, Baez-Ortega, A et al. Somatic mutation rates scale with lifespan across mammals. Nature, 2022.
This article was originally published in the 2021–22 Magdalene College Magazine.
The author is grateful to James Raven and Aude Fitzsimons for their comments on the original manuscript.
